 THE MINERAL VIVIANITE
THE MINERAL VIVIANITE
- Chemistry: Fe3(PO4)2-(H2O)8, Hydrated Iron Phosphate
- Class: Phosphates
- Group: Vivianite
- Uses: only as a mineral specimen
Specimens
PHYSICAL CHARACTERISTICS:
- Color is blue, green and colorless, darkens upon exposure to light.
- Luster is vitreous.
- Transparency crystals are transparent to translucent.
- Crystal System is monoclinic; 2/m
- Crystal Habits include radiating clusters of prismatic, acicular, or fiberous crystals. Also earthy and encrusting masses. Often lines the inside of fossil shells.
- Cleavage is perfect in one direction.
- Fracture is splintery.
- Hardness is 1.5 - 2.
- Specific Gravity is approximately 2.6+ (average for translucent minerals)
- Streak is white or bluish green.
- Associated Minerals are siderite, sphalerite, quartz and some secondary ore deposit minerals.
- Other Characteristics: thin crystals are flexible.
- Notable Occurances include Maryland and Colorado, USA; Russia; Ukraine and England.
- Best Field Indicators are color and reaction to light, flexible crystals and crystal habit.
 Amethyst Galleries' Mineral Gallery MINERALS |

VIVIANITE specimen viv-1
$ 35.00
$ 35.00
Dims: 1-7/8" x 1-7/8" x 1-1/2"
Wt: 23 g
Kertch, Crimean Peninsula, Ukraine
This Vivianite specimen seems to have grown out of a Bauxite host rock. The crystals are a dark bluish green, and very sharp, achieving lengths of about 3/4 inch. This mineral is photosensitive; it should be kept away from light if one does not want the crystals to turn black.

viv-1 ($ 35.00)
Kertch, Crimean Peninsula, Ukraine

VIVIANITE specimen viv-2
$ 30.00
$ 30.00
Dims: 2" x 1-1/4" x 1"
Wt: 22 g
Kertch, Crimean Peninsula, Ukraine
This Viviaite sample has a surprise to it. In between the seemingly Bauxite base and the crystal cluster, there is a layer of calcium carbonate material that is actually a fossil clam shell fragment! The Vivianite cluster is concentrated, with many bladed crystals in a tight formation, and the color is a bluish green that is so dark as to appear black on first glance.

viv-2 ($ 30.00)
Kertch, Crimean Peninsula, Ukraine

VIVIANITE specimen viv-3
$ 48.00
$ 48.00
Dims: 1-3/8" x 1" x 7/8"
Wt: 13.5 g
Morococala, Oruro Deposit, Bolivia
I like simple mineral specimens; this one is no exception. It is thumbnail-sized, and consists of a singular crystal of Vivianite on a siderite matrix. This crystal has no damage whatsoever! It is perfectly shaped in the form of a pseudotetragonal prism with a basal pinacoid termination. It is transparent, and has a splendid dark green color with just a hint of blue! The end opposite the terminaton is exposed, and denotes a clean separation from another crystal, due to the clean, exposed and striated faces. I really like this!

viv-3 ($ 48.00)
Morococala, Oruro Deposit, Bolivia

VIVIANITE specimen viv-4
$ 80.00
$ 80.00
Dims: 3-5/8" x 2-5/8" x 3/4"
Wt: 2.54 oz
Morococala, Oruro Deposit, Bolivia
If you want a big Vivianite crystal, this is for you! The entire specimen is one big crystal! It shows considerable damage, but it is difficult to determine whether or not this damage occurred naturally. Anyhow, it is transparent in some spots, and translucent to opaque in others. There are many areas of striation on the crystal and it has a lovely dark green color. It has a tabular prismatic formation, and one side of it is completely obscured by host rock, where it seems that the crystal was attached. It has a basal pinacoid termination that is slightly damaged and a vitreous luster. It is certainly one of the largest individual crystals that I've seen.

viv-4 ($ 80.00)
Morococala, Oruro Deposit, Bolivia

VIVIANITE specimen viv-5
$ 130.00
$ 130.00
Dims: 3" x 1-1/8" x 1-1/4"
Wt: 2.34 oz
Morococala, Oruro Deposit, Bolivia
This crystal is one of the finer specimens of Vivianite that I've had the pleasure to see. It confuses me, though, because I cannot tell whether the crystal is doubly-terminated or if it is actually a base-to-base penetration twin! Needless to say, it is a single, four-sided monoclinic prism with terminations on each end. These terminations show noticeable damage but are well-formed. The prism faces are all clean with no damage, and show "termination faces" on two opposing sides. Its color is a deep green and is almost transparent in some spots, whereas other places are translucent to opaque. Its luster is vitreous, and it has a small amount of a metallic sulfide matrix attached to it near the center of the prism. There may be some siderite in the matrix rock, too!


viv-5 ($130.00)
Morococala, Oruro Deposit, Bolivia

VIVIANITE specimen viv-6
$ 85.00
$ 85.00
Dims: 2-1/2" x 2-3/8" x 1-1/2"
Wt: 2.71 oz
Morococala, Oruro Deposit, Bolivia
About 7 Vivianite crystals occupy this specimen from the prolific Oruro Deposit, the largest exceeding 1-1/4 inches in length and approaching 1/2 inch in width. The crystals are all a dark blue-green in color and range from near-transparent to opaque in clarity. Their luster is uniformly pearly. There is some noticeable damage to the crystals, but only if one searches for it- the specimen appears flawless upon first glance. These crystals rest on a bed of what I'm pretty sure is siderite- it is shiny, black, and is coated with many tiny botryoidal nodules.

viv-6 ($ 85.00)
Morococala, Oruro Deposit, Bolivia

VIVIANITE specimen viv-7
$ 150.00
$ 150.00
Dims: 2.8" x 0.5" x 0.2"(7.1 x 1.3 x 0.5 cm)
Wt: 2.8 g
Huanuni, Bolivia
Though only 4 Vivianite crystals make up this specimen, they are exceptional. All are in the form of long, six-sided, needle-like prismatic crystals with odd, asymmetrical terminations. The smallest of these crystals measures 0.7"(1.8 cm) long and is the only crystal with noticeable damage, as its termination is missing. All of the Vivianites have a vitreous luster, dark bluish-green coloration, and excellent form. Their edges are somewhat rounded and their faces are slightly striated and "pockmarked", but the crystals are transparent and free of internal flaws. They are splendid, and one could facet some long baguette stones out of the larger crystal. By the way, there is no host rock to speak of.

viv-7 ($150.00)
Huanuni, Bolivia

$3000.00
Dims: 5.8" x 5.2" x 3.4" (14.7 x 13.2 x 8.6 cm)
Wt: 2 lbs., 6.8 oz. (1.100 kg)
Llallagua, Bolivia
This museum-worthy specimen consists of 3 to 5 clusters of large, monoclinic, bladed prismatic Vivianite crystals. They are in generally very good condition, and though there are several visible breakage surfaces, most of these appear to have occurred either before or during the specimen's harvest. The largest discernable blade that I can see has dimensions of 4.3 x 1.6 x 0.3" (10.9 x 4.1 x 0.8 cm). Some of the crystals are quite malformed either due to damage or growing conditions, but most have excellent form with sharp edges and smooth, clean faces. The crystals have a color that ranges from a deep greenish-blue to a deep forest green. Though all seem to have very faint, smoke-like cloudy inclusions, even some of the larger crystals have impressive clarity, and all have a shiny, vitreous luster. There are several patches of a golden, metallic sulfide mineral (I am guessing marcasite) on the specimen, many of which partially cover some of the breakage surfaces. Other than that, there is no other material or host rock on the specimen. It is easily the largest and highest-quality Vivianite specimen that I can remember seeing.


Llallagua, Bolivia

VIVIANITE specimen viv-9
$ 120.00
$ 120.00
Dims: 5.8" x 5.2" x 3.4" (14.7 x 13.2 x 8.6 cm)
Wt: 5.27 oz. (149.6 g)
Tchenomorskiy Mine, Kertch, Krym Peninsula, Crimea, Ukraine
Several clusters of monoclinic, bladed Vivianite crystals rest on the host rock of this specimen. These clusters (and hence, the crystals) do not exceed 1.2" (3.0 cm) in length, and are generally in very good condition, as a few of them show damage and incomplete crystals. All show moderate form, as they are quite intergrown, and their prism and termination faces do not appear to be definite. They have a deep blue to deep green coloration and a pearly luster, and are dimly transparent when viewed in a bright light. They rest upon a very thin, black, botryoidal crust that I would guess is made up of an iron or manganese oxide. This crust rests on the brown, oolitic host rock, which looks an awful lot like bauxite.


viv-9 ($120.00)
Tchenomorskiy Mine, Kertch, Krym Peninsula, Crimea, Ukraine

VIVIANITE specimen viv-10
$ 55.00
$ 55.00
Dims: 1.4" x 1.0" x 0.4" (3.6 x 2.5 x 1.0 cm)
Wt: 6.3 g
Huanuni, Oruro Department, Bolivia
This small specimen consists of a single radiating cluster of thin Vivanite needles. These needles do not exceed 1.4" in length or 2 mm in thickness, and are generally in good condition, though the tips of several of them are obviously missing. They show a pearly luster and are dimly translucent under a bright, halogen light. However, that is enough for one to notice that the entire cluster has a uniform, dark grayish-blue coloration! There is a small amount of an indeterminate, pale brown host rock that is attached to one side of the cluster, but the point from which all of the crystals radiate is free of material. All of the Vivianite specimens that we have seen before this one have been either a pure, dark green or a deep blue-green in color; this is the first that we have seen with a completely blue coloration.

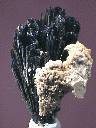
viv-10 ($ 55.00)
Huanuni, Oruro Department, Bolivia

VIVIANITE specimen viv-11
$ 30.00
$ 30.00
Dims: 2.0 x 1.2 x 1.0" (5.1 x 3.0 x 2.5 cm)
Wt: 1.27 oz. (36.1 g)
Kertch, Krym Peninsula, Crimea, Ukraine
Though it is one of our least expensive specimens, this piece pleases me immensely. It consists of a cluster of several small Vivianite prisms that rest in a hollow in a brown-and-white host rock. These crystals are in excellent condition due to the protective nature of the hollow, and are limited by growing space to lengths of 0.4" (1.0 cm) or less. Their monoclinic prismatic form varies from crystal to crystal, but is generally good- their edges are moderately well-defined, and their faces are clean and show a vitreous luster. Though most of them are situated inside the deep and dark hollow, a few exposed crystals show their deep blue-to-green coloration and dim transparence. The aspect of this specimen that makes it so attractive to me is the fact that the host rock is made up of bits of fossil shells trapped in what appears to be bauxite. The hollow in which the Vivianites rest is created by a space between a piece of shell and the bauxite base!

viv-11 ($ 30.00)
Kertch, Krym Peninsula, Crimea, Ukraine

VIVIANITE specimen viv-12
$ 32.00
$ 32.00
Dims: 1.9 x 1.6 x 1.1" (4.8 x 4.1 x 2.8 cm)
Wt: 1.70 oz. (48.3 g)
Tchenomorskiy Mine, Kertch, Krym Peninsula, Crimea, Ukraine
Several radiating sprays of Vivianite crystals fill a hollow in the dull, brown host rock of this thumbnail specimen. These crystals do not exceed 0.8" (2.0 cm) in length but are generally in excellent condition due to the protective nature of their surroundings- only the most exposed crystals are broken or damaged. Their monoclinic prismatic form is somewhat disseminated because of their cramped growing conditions, but is still discernable. The most exposed intact crystal has a rather dull brown coloration and is moderately to dimly translucent, whereas the other crystals are too sheltered to effectively study with respect to clarity. The brown host rock in which they rest contains many nodules and looks very much like bauxite.

viv-12 ($ 32.00)
Tchenomorskiy Mine, Kertch, Krym Peninsula, Crimea, Ukraine

VIVIANITE specimen viv-13
$ 95.00
$ 95.00
Dims: 0.6 x 0.6 x 0.3" (1.5 x 1.5 x 0.8 cm)
Wt: 1.2 g
Chicote Grande Mine, La Paz, Bolivia
A single Vivianite crystal on a pyrite base makes up this small thumbnail specimen. The Vivianite is in pristine condition, showing no damage of any sort, and has dimensions of 0.5 x 0.3 x 0.2" (1.3 x 0.8 x 0.5 cm). Its monoclinic prismatic form is likewise excellent, with very well-defined edges and clean, smooth faces that possess a bright pearly luster. Its color is a moderately dark forest green with a subtle hint of blue, and it is transparent and so clear as to be gem-quality, containing only 2 or 3 very small internal fractures. It rests on a thin crust made up of countless tiny (nearly microscopic) pyrite crystals that have a deep golden color that is tinged with a colorful iridescence and the standard metallic luster. I have placed it in a domed plastic thimble box for display and protection.

viv-13 ($ 95.00)
Chicote Grande Mine, La Paz, Bolivia

VIVIANITE specimen viv-14
$ 135.00
$ 135.00
Dims: 10. x 0.8 x 0.7" (2.5 x 2.0 x 1.8 cm)
Wt: 6.1 g
Chicote Grande Mine, La Paz, Bolivia
At least 4 different Vivianite crystals intersect to form this thumbnail specimen. All are in excellent condition, showing no damage, and achieve dimensions of 0.9 x 0.4 x 0.1" (2.3 x 1.0 x 0.3 cm). Their monoclinic bladed form is very good, with well-defined edges and clean faces that possess a pearly luster. Both deep blue and green colors are visible in each crystal, and all are transparent and impressively clear. They rest on a thin crust of pyrite that acts as a base.


viv-14 ($135.00)
Chicote Grande Mine, La Paz, Bolivia

VIVIANITE specimen viv-15
$ 75.00
$ 75.00
Dims:1.7x0.2x0.1" (4.3x0.5x0.3 cm)
Wt: 0.1oz. (3g)
Huanuni, Oruro Department, Bolivia
This specimen consists of a single flat, tabular crystal of vivianite. This crystal is a lovely dark green in color and is very nearly completely transparent. The termination on this crystal is in very good condition. There is only very minor damage to this specimen.


viv-15 ($ 75.00)
Huanuni, Oruro Department, Bolivia

VIVIANITE specimen viv-17
$ 105.00
$ 105.00
Dims: 5.0x2.8x2.0" (12.6x7.0x5.0 cm)
Wt: 11.5 oz. (325g)
Kertch, Ukraine
This cabinet specimen presents several sprays of dark blue (almost black) vivianite crystals on an unusual matrix. This host rock is composed of hundreds of round nodules glued together by a crumbly clay-like material. Every cavity is filled by vivianite, as was much of the largest cavity in the original rock (where the vivianite is now exposed). Under a loupe, the smallest needles of vivianite are tranparent and blue. This is most easily seen on the crystals which are somewhat hidden and therefore protected from light. Remember that vivianite darkens upon exposure to light, and specimens should be stored in the dark as much as possible.


viv-17 ($105.00)
Kertch, Ukraine

VIVIANITE specimen viv-16
$ 75.00
$ 75.00
Dims: 3.3x2.0x1.3" (8.5x5.2x3.3cm)
Wt: 3.13 oz. (88.6g)
Kertch, Crimea iron deposit, Ukraine
This is specimen is from a cavity which was lined with a crust of vivianite and contains several well-formed sprays of the mineral. Two of the clusters are large and very nice, although the vivianite has darkened due to exposure to light (and will continue to darken further) so that it is nearly black. There is still some transparency, and there are bright blue highlights reflecting from the crystals. Vivianite darkens with exposure to light, and should be kept in a box when not being admired.


viv-16 ($ 75.00)
Kertch, Crimea iron deposit, Ukraine

VIVIANITE specimen viv-18
$ 40.00
$ 40.00
Dims: 2.83x1.69x0.63" (7.2x4.3x1.6cm)
Wt: 2.54oz (72.0g)
Llallagua, Bustillo, Potosi, Bolivia
This specimen is a book of vivianite crystals, or possibly a single crystal that has many parallel cleavage planes (which can result when a tabular crystal of vivianite is flexed, either naturally or by hand). The vivianite, while still having a bluish-green color, has darkened to a dim translucency. Only the thinnest areas show transparency. Two edges seem natural, one face is clearly cleavage, and the other is a mixture of cleavage and splintery fracture. There is a vein of something else penetrating the specimen - it looks like pyrite, but it has weathered to a brown porous material and may indeed be something else.


viv-18 ($ 40.00)
Llallagua, Bustillo, Potosi, Bolivia

VIVIANITE specimen viv-19
$ 25.00
$ 25.00
Dims: 2.77x1.23x0.83" (7.03x3.13x2.12cm)
Wt: 1.27oz (35.9g)
Llallagua, Bustillo, Potosi, Bolivia
While the unique blue color of vivianite is apparent on this specimen, it has darkened to nearly black, especially on the exposed top. Some protected areas on the bottom have an appealing crystal shape and still appear transparent. The crystal is quite fragile, and tiny flakes of vivianite come off during handling, although they don't appear to significantly affect the overall appearance of the specimen. The specimen has several natural faces (not just cleavage planes), and is relatively nice even though it shows some damage.

viv-19 ($ 25.00)
Llallagua, Bustillo, Potosi, Bolivia

VIVIANITE specimen viv-20
$ 60.00
$ 60.00
Dims: 3.10x2.80x1.92" (7.87x7.11x4.87cm)
Wt: 6.28oz (177.7g)
Kerch, Ukraine
This specimen is a brown conglomate host rock (brown balls cemented by a soft brown matrix), with a cavity that has been lined with a thin layer of vivianite, plus many long prismatic crystals of viviantite. These crystals are gray-blue and transparent, but their surface luster, while vitreous, gives them a nearly black opaque appearance. The crystals have a cross section and termination rather like a sword. The optical effect is unusual, in that the crystals appear dark and opaque until you hold them up to a light, then they clearly are transparent. Of course, viviantite darkens with exposure to light, so this specimen should be stored in the dark when not on display.


viv-20 ($ 60.00)
Kerch, Ukraine

VIVIANITE specimen viv-21
$ 43.00
$ 43.00
Dims: 2.56x1.86x1.36" (6.49x4.73x3.45cm)
Wt: 3.62oz (102.5g)
Kertch peninsula, Crimea Oblast', Ukraine
One bivalve shell on this fossiliferous rock has been broken open to reveal two dozen vivianite crystals hidden within. While the surfaces are dark, nearly black, they are transparent enough to show the unique blue color of vivianite crystals. When held to the light all of the crystals appear transparent and have this blue to blue-green color, so the black is a surface effect. Of course, vivianite does darken upon exposure to light, so it should be stored in a box or other dark area when it is not being viewed.


viv-21 ($ 43.00)
Kertch peninsula, Crimea Oblast', Ukraine