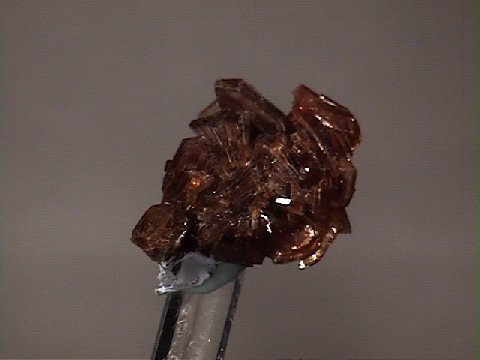 THE MINERAL HEULANDITE
THE MINERAL HEULANDITE
- Chemistry: (Ca, Na)2 - 3Al3(Al, Si)2Si13O36 - 12H2O, Hydrated Calcium Sodium Aluminum Silicate
- Class: Silicates
- Subclass: Tectosilicates
- Group: Zeolites
- Zeolite Family: Heulandite
- Uses: As chemical filters and as mineral specimens.
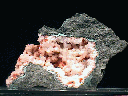
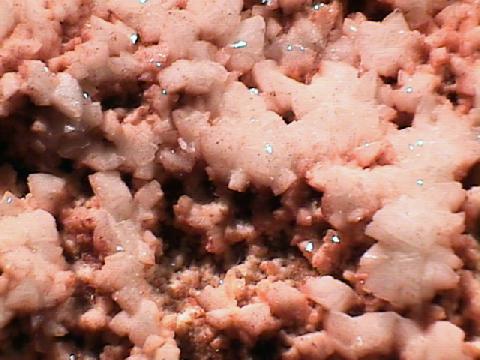
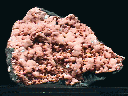
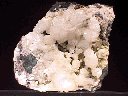
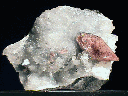
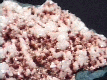
Specimens
Heulandite's structure is sheet-like. Although still a true tectosilicate where every oxygen is connected to either a silicon or an aluminum ion (at a ratio of [Al + Si]/O = 1/2) and the structure is a framework, there still is a sheet-like structural organization. The sheets are connected to each other by a few bonds that are relatively widely separated from each other. The sheets contain open rings of alternating eight and ten sides. These rings stack together from sheet to sheet to form channels throughout the crystal structure. The size of these channels controls the size of the molecules or ions that can pass through them and therefore a zeolite like heulandite can act as a chemical sieve, allowing some ions to pass through while blocking others. A zeolite can be thought of in terms of a house, where the structure of the house (the doors, windows, walls and roof) is the zeolite while the furniture and people are the water, ammonia and other molecules and ions that can pass in and out of the structure. In the case of heulandite, the sheet-like structure could be analogous to the floors of a high-rise office building with only a few braces between the floors. Heulandite's sheet-like structure produces the prominent pinacoid faces, the perfect cleavage and the unique luster on those faces.
Heulandite shares its structure with the closely related mineral clinoptilolite. Clinoptilolite in fact is considered by some experts to be a high potassium, high silica variety of heulandite. But for now it is considered a distinct species. Natural heulandite specimens can have significant amounts of strontium, potassium, magnesium and barium in their chemical makeup. At times the formula is written to show all these ions and varieties that are enriched in these ions are referred to as strontium rich heulandite for example.
PHYSICAL CHARACTERISTICS:
- Color is colorless, white, gray, green, pink, yellow, red, brown and black.
- Luster is vitreous to pearly on the most prominent pinacoid face and on cleavage surfaces.
- Transparency: Crystals are transparent but most commonly translucent.
- Crystal System is monoclinic; 2/m
- Crystal Habits include blocky crystals described as diamond-shaped, trapezoidal and old fashioned coffin shape with the two faces of a pinacoid usually prominent. Crystals are often modified by secondary faces with pairs of triangular faces very common. Prismatic and acicular forms are also known and are difficult to identify as heulandite. In aggregate specimens this face can be oriented upward producing a crust of curved pearly faces or it can be oriented to the side where the tops of the crystals jut out like the roof tops of a suburban community. Crystals can be simple or complexly modified by a variety of prism and pinacoid faces.
- Cleavage is perfect in one direction parellel to the prominent pinacoid face.
- Fracture is uneven.
- Hardness is 3.5 - 4, maybe softer on cleavage surfaces.
- Specific Gravity is approximately 2.1 - 2.3 (very light due to the open channels and high water content).
- Streak is white.
- Associated Minerals are extensive and include quartz,
calcite,
apophyllite,
barite,
pyrite,
prehnite,
pollucite , tourmaline, scolecite, analcime, chabazite,ferrierite , mordenite, laumontite, natrolite, stilbite and other zeolites. - Notable Occurrences are wide spread and include Poona and Nasik Districts, India; Iceland; New Jersey; Washington; Boron, California; Patterson, New Jersey; Arizona and Oregon, USA; Nova Scotia and Quebec, Canada; Iran; Sardinia, Italy; Rio Grande do Sul and Goias, Brazil; New South Wales and Phillip Island, Victoria, Australia; New Zealand; Kola Peninsula, Russia; Scotland and Rhone Valley, Switzerland.
- Best Field Indicators are crystal habit, density, cleavage, luster and associations.