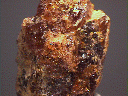THE
MINERAL PARISITE
THE
MINERAL PARISITE
- Chemistry: Ca(Ce, La, Nd)2(CO3)3F2 , Calcium Cerium Lanthanum Neodymium Carbonate Fluoride.
- Class: Carbonates
- Groups: Rare earth carbonates and Bastnasite.
- Uses: As a minor ore of cerium and other rare earth metals and as mineral specimens.
Specimens
Parisite, which is named for the owner of the mine (J.J. Paris) at which parisite was discovered, is one of a
few rare earth carbonate minerals.
Some of the other more common rare earth carbonates are
Parisite is closely related to three other distinct minerals;
synchysite,
bastnasite
and
Parisite is only found as small crystals and can be used for micromounts. Some crystals have been reported to have been cut as gems, but normally the crystals of parisite are too small and cloudy to make good gemstones. Parisite crystals are found in carbonatites, granite pegmatites and alkaline syenites and the hydrothermal deposits associated with them. Parisite is a significant ore at the bastnasite mines of Mountain Pass, California.
PHYSICAL CHARACTERISTICS:
- Color is white, tan, yellow-brown, reddish-brown, brown, yellow and pale pink.
- Luster is vitreous, resinous to dull.
- Transparency: Crystals are translucent to more rarely transparent.
- Crystal System is trigonal.
- Crystal Habits include small acicular, platy or thin tabular crystals that form rosettes. Also known to form double hexagonal pyramids and rhombohedrons. Often intergrown in a lamellar fashion with synchysite, rontgenite and bastnasite.
- Cleavage is fair to good in one direction (basal).
- Fracture is subconchoidal to splintery.
- Hardness is typically around 4.5 but will vary from 4 to 5.
- Specific Gravity is approximately 4.2 to 4.3 (well above average)
- Streak is white to yellowish white.
- Other Characteristics: Only slightly soluble in nitric acid, a strong basal parting is also sometimes seen and crystals are typically striated parallel to the basal face.
- Associated Minerals are extensive and include
bastnasite,
rontgenite-(Ce) , synchysite, gold, fluorite,sahamalite-(Ce) , dolomite, barite,allanite , aegirine, calcite,ancylite , albite, pectolite, microcline, riebeckite,cerite , rhodochrosite, sphene, ankerite, biotite, monazite and xenotime. - Notable Occurrences include the type locality in the Muzo emerald deposit in the Muzo district, Bogota, Colombia.
Other localities include the Mountain Pass, San Bernardino, California; Quincy, Mass.; Mohave County, Arizona and Mineral and Ravelli Counties, Montana, USA;
Narsarsuk , Greenland; the mines of Mont Saint-Hilaire, St. Honore and Gatineau, Quebec and the Thor Lake deposits, Northwest Territories, Canada;langesundsfjord area, Norway and the Weishan deposit, Weishan Lake district, Shandong Province, China and the type locality for parisite-(Nd) that being Bayan Obo, inner Mongolia, China. - Best Field Indicators: crystal habit, color, cleavage, density, luster, low solubility in nitric acid and locality.