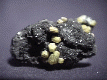 THE ETTRINGITE GROUP OF MINERALS
THE ETTRINGITE GROUP OF MINERALS
The Ettringite Group is composed of hydrated calcium sulfate hydroxide minerals with a common
trigonal structure.
It is named for its most common member,
ettringite, which is actually a rather uncommon mineral.
The general formula for this group is Ca
6X
2(SO
4)
3(O, OH)
12 - 26H
2O.
The X can be either aluminum, chromium, iron, manganese or silicon.
The SO
4 ion group can be partially replaced by the rare B(OH)
4 ion group as in a couple of the members of this group
sturmanite and
charlesite.
The SO
4 ion group can also be partially replaced by carbonate ions as in
thaumasite and
jouravskite.
In
ettringite and
bentorite, these types of substitutions are minimal.
Thaumasite is sometimes classified as a
silicate because of its silicon content, but it lacks the SiO
4 tetrahedrons required for inclusion in that class.

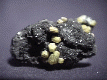 THE ETTRINGITE GROUP OF MINERALS
THE ETTRINGITE GROUP OF MINERALS