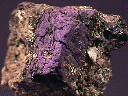- Chemistry: Zn2AsO4(OH), Zinc Arsenate Hydroxide
- Class: Phosphate Class
- Subclass: Arsenates
- Uses: Only as mineral specimens
Specimens
Paradamite is dimorphous with a famous arsenic mineral, namely adamite. Dimorphous means that the two minerals have the same formula, but different structures (di means two; morphous means shape). Paradamite's different structure produces only slight differences in physical properties. Most obvious however is the difference in crystal forms. Adamite's typical form is wedge shaped prismatic crystals with diamond-shaped cross-sections. Paradamite's form is more tabular in character and very different from adamite's. Although their names are similar and their chemistry is the same; paradamite and adamite are absolutely distinct minerals.
Curiously, paradamite is associated with adamite as well as other common oxidized arsenic minerals. It is curious because often dimorphic minerals are not directly associated. The diverse conditions required to produce a different structure from the same chemistry are often so radically different that the minerals are not found together. However if the conditions required to produce the two minerals are only slightly different; then the two minerals can be found together and sometimes grade into each other. What those exact conditions are, and how much they must change to produce the dimorph is of great interest to mineralogists and petrologists.
PHYSICAL CHARACTERISTICS:
- Color is pale yellow
- Luster is vitreous.
- Transparency: Crystals are transparent to translucent.
- Crystal System is triclinic; bar 1.
- Crystal Habits include rounded tabular crystals, usually aggregated.
- Cleavage is perfect.
- Fracture is uneven.
- Hardness is 3.5.
- Specific Gravity is approximately 4.5 - 4.6 (heavy for translucent minerals)
- Streak is white.
- Other Characteristics: Some specimens are fluorescent yellow (adamite usually fluoreses green) under shortwave UV light.
- Associated Minerals include adamite, legrandite, limonite, smithsonite, austinite, mimetite and other zinc oxidation zone minerals.
- Notable Occurrence is limited to the famous mines of Mapimi, Durango, Mexico.
- Best Field Indicators are crystal habit, color, density, fluorescence and associations.